Carotid–femoral pulse wave velocity: Impact of different arterial path length measurements
- DOI
- 10.1016/j.artres.2009.11.001How to use a DOI?
- Keywords
- Arterial stiffness; Applanation tonometory; Carotid artery
- Abstract
Background: Carotid–femoral pulse wave velocity (PWV) is the most established index of arterial stiffness. Yet there is no consensus on the methodology in regard to the arterial path length measurements conducted on the body surface. Currently, it is not known to what extent the differences in the arterial path length measurements affect absolute PWV values.
Methods: Two hundred fifty apparently healthy adults (127 men and 123 women, 19–79 years) were studied. Carotid–femoral PWV was calculated using (1) the straight distance between carotid and femoral sites (PWVcar–fem), (2) the straight distance between suprasternal notch and femoral site minus carotid arterial length (PWV(ssn–fem)−(ssn–car)), (3) the straight distance between carotid and femoral sites minus carotid arterial length (PWV(car–fem)−(ssn–car)), and (4) the combined distance from suprasternal notch to the umbilicus and from the umbilicus to femoral site minus carotid arterial length (PWV(ssn–umb–fem)−(ssn–car)).
Results: All the calculated PWV were significantly correlated with each other (r = 0.966–0.995). PWVs accounting for carotid arterial length were 16–31% lower than PWVcar–fem. PWVcar–fem value of 12 m/s corresponded to 8.3 m/s for PWV(ssn–fem)−(ssn–car), 10.0 m/s for PWV(car–fem)−(ssn–car), and 8.9 m/s for PWV(ssn–umb–fem)−(ssn–car).
Conclusion: Different body surface measurements used to estimate arterial path length would produce substantial variations in absolute PWV values.
- Copyright
- © 2009 Association for Research into Arterial Structure and Physiology. Published by Elsevier B.V. All rights reserved.
- Open Access
- This is an open access article distributed under the CC BY-NC license.
Introduction
Clinical importance of arterial stiffness has been well documented.1,2 Most of the landmark studies in this area have used carotid–femoral pulse wave velocity (PWV) to quantify arterial stiffness.3–9 In order to calculate PWV, arterial path length is estimated using the distance measured on the body surface. Many of the aforementioned classic studies have assessed aortic PWV using the straight distance between the two recording sites (the carotid and femoral artery).5,7,9 Based on mounting evidences, European Society of Hypertension (ESH) and European Society of Cardiology (ESC) published guidelines declaring a borderline or threshold value of aortic PWV (>12 m/s) that should be used to stratify cardiovascular risks in hypertensive patients.10 However, this particular value is applicable only to aortic PWV obtained using the straight distance between the carotid and femoral artery. Increasing number of investigators have started using the length obtained by subtracting the distance between the suprasternal notch to the carotid site from main path length (e.g., suprasternum–femoral or carotid–femoral) in an attempt to account for the opposite pulse transition toward the carotid artery.4,11–13 PWV values reported in these studies appear to be much lower than those reported in the classic studies. Recently, Rajzer et al.14 compared the values of carotid–femoral PWV obtained using the Complior and the SphygmoCor and reported that PWV measured with the former method was 25% higher compared with that using the latter method. This was attributed to a difference in the arterial path length measurement required in the Complior and the SphygmoCor. There have been no studies that comprehensively assessed the impact of different body surface measurements on absolute PWV values. Additionally, it is not known what specific PWV values would correspond to the threshold value of 12 m/s recorded on the carotid–femoral PWV if a different method was used to estimate arterial path length.
Accordingly, the purpose of this study was to assess the impact of a variety of arterial path length measurements on carotid–femoral PWV value.
Methods
We studied a total of 250 adults (127 men and 123 women, 19–79 years). Subjects were free of overt cardiovascular disease as assessed by health status questionnaire. A total of 27 patients were taking prescribed antihypertensive (n = 24), cholesterol lowering (n = 6), diabetic (n = 4), and other (e.g., thyroid hormone, anticoagulation) (n = 8) medications. All potential risks and procedures of the study were explained to the subjects, who gave their written informed consent to participate in the study. This study was reviewed and approved by the Institutional Review Boards at the National Institute of Advanced Industrial Science and Technology and the University of Texas at Austin.
All measurements were performed after 3 h fasting and an abstinence of caffeine. Subjects were studied under supine resting conditions in a quiet, temperature-controlled room (22–24 °C). Carotid and femoral artery pulse waves were recorded by arterial applanation tonometry incorporating an array of 15 micro-piezoresistive transducers placed on the carotid and femoral arteries (VP-2000, Colin Medical, San Antonio, TX) as previously described in detail.15 The time delay (ΔT) was measured automatically with the foot-to-foot method. Using a customized segmometer specifically designed for PWV studies (Rosscraft Anthropometric Calipers, Surray, Canada, Fig. 1), the following body surface lengths typically assessed in PWV studies were measured:
where “car–fem” is the straight distance between the carotid (car) and the femoral (fem) artery recording sites, “ssn–fem” is the straight distance between the suprasternal notch (ssn) and the femoral artery site, “ssn–car” is the straight distance between the suprasternal notch and the carotid artery site, “ssn–umb–fem” is the combined distance from the suprasternal notch to the umbilicus (umb) and from the umbilicus to the femoral site. Carotid–femoral PWV was subsequently calculated from the time delay and a variety of arterial path lengths measured with the body surface measurement (PWVcar–fem, PWV(car–fem)−(ssn–car), PWV(ssn–fem)−(ssn–car), and PWV(ssn–umb–fem)−(ssn–car)). In our previous studies, when body surface arterial path lengths were measured in triplicate, coefficients of variations were very small: 0.53% for car–fem, 0.76% for (car–fem)−(ssn–car), 0.80% for (ssn–fem)−(ssn–car), and 0.84% for (ssn–umb–fem)−(ssn–car).
A customized segmometer (top: slider indicators; bottom: entire image). Segmometer consists of a flexible metal tape like a retractable carpenter’s tape and two slider indicators.
Brachial blood pressure and heart rate were measured with oscillometric pressure sensor cuffs and electrocardiograms (VP-2000, Colin Medical, San Antonio, TX).
Simple Pearson product moment correlation and regression analyses were performed to determine relations of interests. To identify significant differences among mean values of PWV, ANOVA and the subsequent post hoc testing (Newman–Keuls method) were performed. All data are reported as mean ± SD. Statistical significance was set a priori at P < 0.05.
Results
Subject characteristics are described in Table 1. Arterial path length of car–fem was significantly longer than the other arterial path lengths (Table 2). Consequently, PWVcar–fem was 16–31% greater (P <0.0001) than PWV calculated with the other body surface measurements (Table 2). PWV accounting for carotid arterial length demonstrated strong linear correlations with PWVcar–fem (r = 0.966–0.995, all P < 0.0001, Fig. 2). The extrapolation procedure using the regression equations reveal that the clinical diagnostic threshold of PWVcar–fem of 12 m/s proposed in the ESH and ESC guidelines 10 corresponded to 10.0 m/s for PWV(car–fem)−(ssn–car), 8.3 m/s for PWV(ssn–fem)−(ssn–car), and 8.9 m/s for PWV(ssn–umb–fem)−(ssn–car).
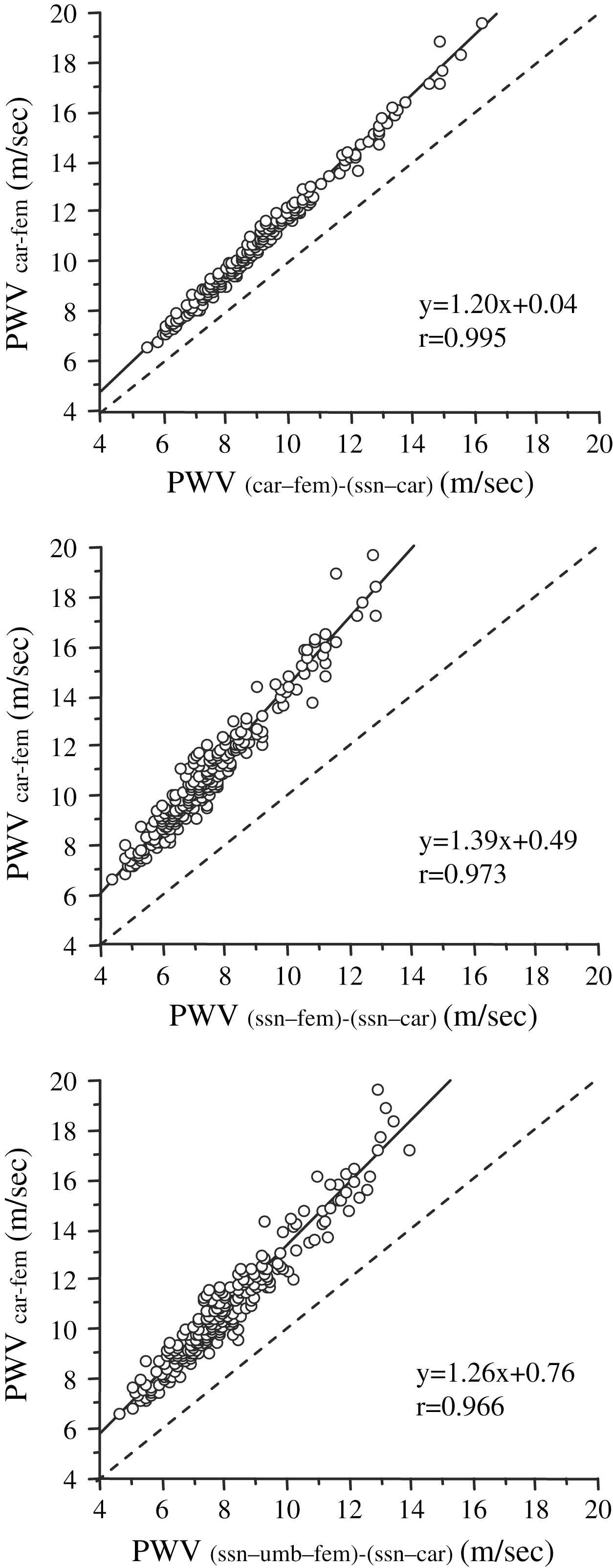
Comparisons of carotid–femoral pulse wave velocity (PWV) calculated using the straight distance between carotid and femoral arterial pressure recording sites (PWVcar–fem) to PWV using arterial path lengths subtracting carotid length from the straight distance between suprasternal notch and femoral site (PWV(ssn–fem)−(ssn–car)), the straight distance between carotid and femoral sites (PWV(car–fem)−(ssn–car)), and the combined distance from suprasternal notch to the umbilicus and from the umbilicus to femoral site (PWV(ssn–umb–fem)−(ssn–car)). Broken lines present the identity line (y = x).
| Male/female, n | 127/123 |
| Age, year | 53 ± 16 |
| Height, cm | 163 ± 9 |
| Body mass, kg | 61 ± 12 |
| Body mass index, kg/m2 | 23 ± 3 |
| Heart rate, bpm | 61 ± 12 |
| Systolic blood pressure, mmHg | 123 ± 16 |
| Diastolic blood pressure, mmHg | 75 ± 11 |
Data are mean ± SD.
Selected subjects’ characteristics.
| Method | Arterial path length, mm | PWV, cm/s |
|---|---|---|
| car–fem | 621 ± 37 | 1081 ± 238 |
| (car–fem)−(ssn–car) | 518 ± 31* | 901 ± 198* |
| (ssn–fem)−(ssn–car) | 427 ± 29*†‡ | 743 ± 166*†‡ |
| (ssn–umb–fem)−(ssn–car) | 458 ± 30*† | 798 ± 182*† |
Data are mean ± SD. “car–fem” is the straight distance between the carotid (car) and the femoral (fem) artery recording sites, “ssn–fem” is the straight distance between the suprasternal notch (ssn) and the femoral artery site, “ssn–car” is the straight distance between the suprasternal notch and the carotid artery site, “ssn–umb–fem” is the combined distances from the suprasternal notch to the umbilicus (umb) and from the umbilicus to the femoral site.
P < 0.0001 vs. car–fem.
P < 0.0001 vs. (car–fem)−(ssn–car).
P < 0.0001 vs. (ssn–umb–fem)−(ssn–car).
Mean arterial path lengths and carotid–femoral pulse wave velocity (PWV).
Among all the subjects studied, a total of 61 subjects had >12 m/s of PWVcar–fem. Threshold values of PWV(car–fem)−(ssn–car) (e.g. 10.0 m/s), PWV(ssn–fem)−(ssn–car) (e.g. 8.3 m/s), and PWV(ssn–umb–fem)−(ssn–car) (e.g. 8.9 m/s) detected 95% (n = 58/61), 92% (n = 56/61), and 87% (n = 53/61) of these cases.
Discussion
Aortic PWV is widely considered the most established index of arterial stiffness.2 Yet there has been no standardization or consensus in terms of how the arterial path length required for PWV should be measured. Currently, different investigators use a variety of methodologies to estimate the arterial path length between the carotid and femoral arteries. In large epidemiological studies from France that yielded the most clinically significant findings on aortic PWV,5,7,9 the straight distance between the carotid and femoral arteries was used. Based primarily on these study findings, the European Society of Hypertension and European Society of Cardiology recently stated that a borderline or threshold value of aortic PWV (>12 m/s) should be used to stratify cardiovascular risks in hypertensive patients.10 Arterial path length measurements-dependent threshold PWV may become a necessity if arterial stiffness measures were to be fully integrated into routine clinical settings. However, other investigations, including population-based large sample-size studies (i.e., Framingham Heart Study, Anglo-Cardiff Collaborative Trial) utilized the subtraction of the carotid artery length from the suprasternal notch to femoral straight distance in order to account for the pulse traveling in the opposite direction.4,11–13 Due to the use of different methodologies to estimate arterial length, inter-study comparisons of aortic PWV have been difficult to perform.
To the best of our knowledge, the present study is the first comprehensive analysis to compare carotid–femoral PWV using a variety of arterial path lengths. PWV values accounting for carotid arterial length (e.g., PWV(car–fem)−(ssn–car), PWV(ssn–fem)−(ssn–car), and PWV(ssn–umb–fem)−(ssn–car)) were 16–31% lower than PWV calculated by the straight distance between carotid and femoral sites (e.g., PWVcar–fem). Interestingly, correlational and regressional analyses depicted extremely strong associations between PWVs obtained with different arterial path length estimations. Although the deviation of correlation seemed to be larger with higher PWVs, greater deviations from a line of identity might be due simply to a mathematical artifact as a greater PWV is associated with a greater arterial path length and/or shorter pulse transition time and would be affected by the conversion to a greater extent. Importantly, equations obtained in the regressional analyses allowed us to compare one specific PWV value (12 m/s) with other PWV values obtained using a different arterial path length estimation. Using this approach, PWVcar–fem value of 12 m/s proposed by the ESH and ESC10 corresponded to 10.0 m/s for PWV(car–fem)−(ssn–car), 8.3 m/s for PWV(ssn–fem)−(ssn–car), and 8.9 m/s for PWV(ssn–umb–fem)−(ssn–car). These specific values could detect 89–95% of cases that PWVcar–fem was higher than 12 m/s.
The results of the present study indicate that a choice of arterial path length measurements elicits markedly different PWV values. It may be reasonable to suggest that it is about time to standardize the measurement of arterial path length measurement required for the pulse wave velocity. The estimation of arterial path length based on the body surface distance seems very arbitrary, and it was not clear which methodology should be used. We recently measured the aortic path lengths directly by the 3-dimensional transverse magnetic resonance image (MRI) arterial tracing in 256 apparently healthy adults.16 We found that PWV calculated with the straight distance between carotid and femoral sites 26% overestimated the actual arterial path length measured on the MRI. In contrast, PWV employing the procedure subtracting carotid length from the straight distance between carotid and femoral sites had a higher linearity and smaller errors (r = 0.97 and 7% of mean difference vs. the actual aortic PWV) compared with PWVs calculated with the other superficial measurement.16 These results suggest that PWV(car–fem)−(ssn–car) would be an ideal candidate to be standardized for. However, Weber et al.17 reported that “suprasternal notch–femoral minus suprasternal notch–carotid distance” (e.g., PWV(ssn–fem)−(ssn–car)) showed the best agreement with invasive catheter-based data. A major reason for the discrepancy may be that Weber et al. measured the distance and transit time from the aortic arch to aortic bifurcation and compared it with the transit time obtained in carotid–femoral PWV. Even though the distance may reflect the actual aortic length, such methodology may underestimate the measure of carotid–femoral PWV because “muscular” iliac artery, which is included in the transit time for cfPWV, is totally ignored in the catheter-based calculations that Weber et al. used in their study.17
In summary, the present study was performed with the main intention to compare aortic (carotid–femoral) PWV values obtained with a variety of the arterial length path estimation methods. The salient finding is although PWV obtained from the carotid–femoral straight distance (e.g., PWVcar–fem) strongly correlated with other PWV values, the use of different arterial length estimation produces markedly divergent PWV values. These results would suggest an urgent need to standardize the carotid–femoral PWV.
Conflicts of interest
None.
Funding sources
Special Coordination Funds of the Japanese Ministry of Education, Culture, Sports, Science, and Technology (16700499), JSPS Postdoctoral Fellowships for Research Abroad, NIH award AG20966.
Acknowledgements
This study was supported in part by the Special Coordination Funds of the Japanese Ministry of Education, Culture, Sports, Science, and Technology (16700499), JSPS Postdoctoral Fellowships for Research Abroad, and NIH award AG20966.
References
Cite this article
TY - JOUR AU - Jun Sugawara AU - Koichiro Hayashi AU - Takashi Yokoi AU - Hirofumi Tanaka PY - 2009 DA - 2009/12/04 TI - Carotid–femoral pulse wave velocity: Impact of different arterial path length measurements JO - Artery Research SP - 27 EP - 31 VL - 4 IS - 1 SN - 1876-4401 UR - https://doi.org/10.1016/j.artres.2009.11.001 DO - 10.1016/j.artres.2009.11.001 ID - Sugawara2009 ER -
