Evaluation of tuberculosis public health surveillance, Al-Madinah province, Kingdom of Saudi Arabia, 2012
- DOI
- 10.1016/j.jegh.2015.04.005How to use a DOI?
- Keywords
- Public health surveillance; Tuberculosis; Surveillance evaluation; Program evaluation; Saudi Arabia
- Abstract
The objective of the study is to evaluate the quality of the data, the sensitivity of the surveillance, and the completeness of identification and investigation of tuberculosis (TB) patient’s contacts. The study covered the TB surveillance program in Al-Madinah province in 2011. First, we reviewed all the notifications, treatment cards, and register books, as well as monthly and quarterly reports, for completeness and accuracy of data. Then, we searched for the missed cases that were not reported. Finally, we reviewed all the patients’ household contacts’ reports to assess the degree of completion of identification and investigation. There were 444 cases detected during the study period; only 200 cases were reported. The sensitivity of the TB surveillance system was 45%. Among the 200 reported cases, the results revealed high completeness rates for demographic and disease data and low completeness rates for the test result fields. The contact identification and investigation showed that 34.4% of smear-positive cases’ contacts were not identified. Only 67% of identified contacts were investigated. The review of hospital records and lab registers showed that 244 cases were not reported. In conclusion, the TB surveillance system has several areas that need improvement.
- Copyright
- © 2015 Ministry of Health, Saudi Arabia. Published by Elsevier Ltd.
- Open Access
- This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/)
1. Introduction
Tuberculosis (TB) is a significant global health problem. Among infectious diseases, TB is the second leading cause of death globally and the single most infectious killer among youth and adults [1]. Currently, one-third of the world’s population is infected with Mycobacterium tuberculosis [2]. Not all TB cases are identified, and some that are identified are not treated. Untreated active TB patients can infect 10–15 additional persons annually [3].
The Kingdom of Saudi Arabia (KSA) does not have a high TB burden, yet it faces real challenges in controlling and preventing TB due to its huge number of pilgrims and migrant workers. In 2011, there were 6200 total cases in KSA, of which 4900 were incident cases. In the same period, there were 1100 deaths due to TB. In addition, there were 110 cases of TB and HIV co-infections. According to WHO, 80% of TB cases were detected, and most of the new cases were smear-positive, the most contagious type. WHO estimated that MDR-TB comprised 1.8% of all TB cases. The prevalence and mortality rates over the last 10 years have remained almost unchanged [4]. In the Madinah province, the annual incidence rate of TB cases was 8.6–9.6 per 100,000. The TB surveillance program encompasses the whole province population, which totals 1,858,000 persons.
KSA, like many countries in the world, has a TB surveillance program, which is responsible for data collection, analysis, and feedback, as well as the implementation of actions. The quality of data that are collected by surveillance is a critical attribute. More accurate and complete data increase the usefulness and utilization of information. High detection rates coupled with accurate and complete data help health officials understand trends of diseases in a country and their characteristics in addition to disease burden. The identification of new TB cases among contacts is one of the key steps in prevention. Contacts are defined by KSA’s TB surveillance system as those who share the same home as the patient. A strong surveillance system helps decision makers establish and evaluate different interventions for prevention and control. Further, it assures appropriate medical therapy and follow-up. Beyond that, data can be used to make comparisons between regions, as well as other countries.
The National Tuberculosis Control Committee in KSA was established in 1992 to implement a program throughout the country. They launched the public health surveillance (PHS) and created a manual. When any suspected TB cases are identified in any health care facility, they will be referred to the hospitals for confirmation by sputum smear microscopy, mycobacterial culture, radiography and histopathology. Those with pulmonary TB and sputum smear positive results are admitted to the hospitals for 2 months and their household contacts are investigated for TB. When hospital admission is not required, the hospital or primary health care center will track the case and ensure compliance to the treatment. Due to TB’s public health importance, prevention and treatment related services for TB patients and their contacts are administered through a MOH team regardless of patients’ citizenship or reason for visiting the kingdom.
PHS consists of multiple, consecutive steps. The suspected case is registered in a special logbook and referred for confirmation to the nearest hospital by the physicians. After confirmation, the case is reported to the hospital TB coordinator who is responsible for notification of the regional coordinator. All patients’ lab results and treatment outcomes will be sent to the regional TB coordinator. The regional TB coordinator (1) sends the public health team from the nearest primary health care center to identify and investigate the patient’s household contacts, (2) gives the patient a unique number and sends this number to the hospital to label the patient’s treatment card, and (3) monitors the defaulted cases and persuades them to complete their treatment. Monthly, regional coordinators submit a report of the new and relapsed cases as well as the lab results and treatment outcomes to the central unit [5–7].
To improve PHS, it is essential to evaluate it regularly to ensure that the disease is monitored efficiently and effectively. Different PHS attributes can be assessed depending on the purpose of the program and the aim of the evaluation. The evaluation methods should highlight the program’s substantial features since the PHS programs differ in their purposes, objectives, methods, and target populations [8]. After each evaluation, the recommendations should be adopted to improve the quality, efficiency, and usefulness of the program.
2. Methods
This study was conducted in Al-Madinah, KSA, during the summer of 2012 in order to evaluate Al-Madinah’s TB PHS in 2011. Evaluation was based on all new TB cases diagnosed between Jan 1, 2011, and Dec 31, 2011. We reviewed register books from the three laboratories in Al-Madinah, the admission reports and patients’ diagnoses in hospital records, and the register books in the infection control or public health departments; all confirmed positive cases during the same period were compared with the regionally-reported cases to identify the ones that were missed. Because the system does not require reporting of any cases that are only in the country temporarily, Hajj- and Omrah-labeled cases (i.e., religious pilgrims) were excluded.
All notifications sent from any health facilities to the regional coordinator were examined to assess external completeness. Patient demographic data, contact information, signs and symptoms of the disease, past history, disease data, lab results, HIV test result and reporter data were reviewed.
Due to the highly infectious nature of TB, all of the contact investigations were reviewed. The TB program requires investigation of pulmonary TB patients and their contacts with positive sputum smears. Two steps are associated with contact investigation. The epidemiologist in the nearest primary health center identifies the contacts. Then, those contacts are tested.
To describe the quality (internal completeness) of data, monthly reports sent to the national coordinator were compared with patient treatment cards. We compared the lab results, disease data, treatment plan, and outcome reflected in the monthly reports sent to the MOH and those in the coordinator’s register books.
3. Results
The review and comparison of the hospital records and lab registers with reported cases showed 244 confirmed cases that were not reported to the coordinator, which indicates that the TB surveillance program’s sensitivity was 45%. Among the non-reported cases, 213 (87.3%) were confirmed TB positive by labs. The remaining 31 positive cases (12.7%) were discovered via hospital admission records (Fig. 2).
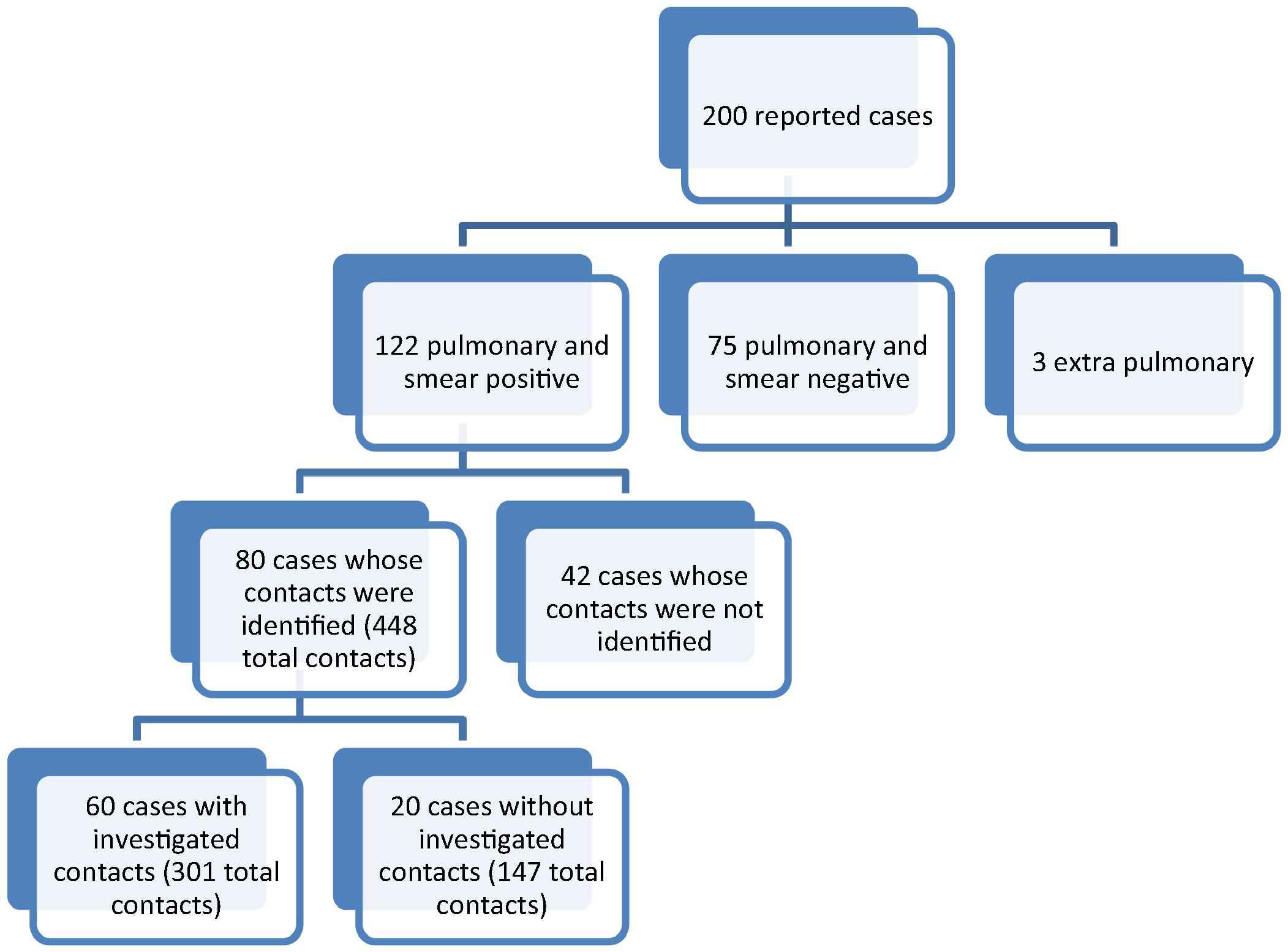
The identification and investigation of the contacts in 2011, Saudi Arabia, Al-Madinah province.
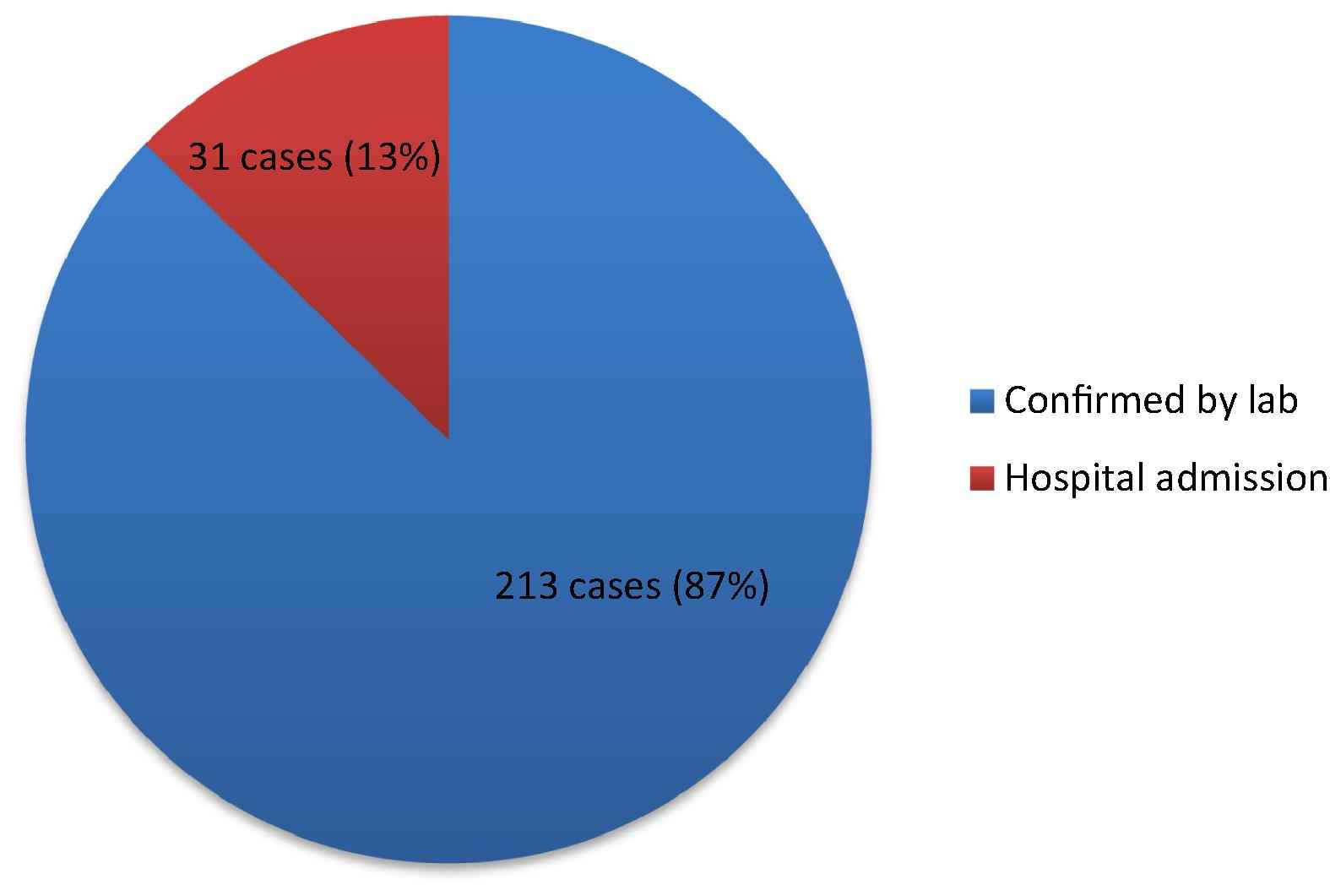
The distribution of missed cases in 2011, Saudi Arabia, Al-Madinah province.
Demographic data were mostly complete (Table 1), ranging from 91.5% to 100%. The lowest external completeness was the identification (ID) field in which out of total 200 cases, 17 were missing (91.5% external completeness). The disease section showed the highest rate of completeness, with 97.5% for signs and symptoms and 94.5% for patient classification and treatment plan.
| Category | Issue | Missed | Completeness rate (%)* |
|---|---|---|---|
| Demographic data | Name | 0 | 100 |
| Nationality | 0 | 100 | |
| Age | 1 | 99.5 | |
| Gender | 3 | 98.5 | |
| ID | 17 | 91.5 | |
| Contact information | Patient telephone number | 21 | 89.5 |
| Disease data | Sign and symptoms | 5 | 97.5 |
| Patient’s classification | 11 | 94.5 | |
| Treatment plan | 11 | 94.5 | |
| Past history | 12 | 94 | |
| Site of the disease | 18 | 91 | |
| Lab test results | Sputum smear | 40 | 80 |
| Chest X-ray | 41 | 79.5 | |
| Tuberculin test | 108 | 46 | |
| Sputum culture | 137 | 31.5 | |
| HIV test | 150 | 25 | |
| Administration data | Admission date | 9 | 95.5% |
| Hospital name | 31 | 84.5 | |
| Doctor name or signature | 41 | 79.5 |
The completeness rate is out of 200 reported cases.
Completeness rate of different categories on TB notification forms, 2011, Saudi Arabia, Al-Madinah province.
Lab test results showed the lowest completeness rates: sputum smear and chest X-ray had 40 blank fields (80% external completeness). In addition, there were 108 missing fields in the category of tuberculin test and 137 in the sputum culture field. The poorest external completeness was seen in the HIV test result fields, 150 of which were left blank.
Administrative data and contact information completeness ranged from 79.5% to 95.5%. Doctor’s name or signature showed 79.5% completeness. The hospital name field was completed in 84.5% of the reports and patients contact information showed an 89.5% completeness rate. The admission data were completed in 95.5% of the reports.
The household contact identification and investigation showed that 80 case contacts were identified (Fig. 1). Those cases with identified contacts had a total of 448 contacts with a median number of 5 contacts per patient. Among the patients whose contacts were identified, 60 patients’ contacts (a total of 301 contacts) were investigated. The 120 cases whose contacts were not identified were distributed as such: 42 cases (35%) were smear-positive, 75 cases (62.5%) were smear negative and 3 cases (2.5%) were extra-pulmonary.
There were 145 patient treatment cards available out of 200 (Table 2). After reviewing 12 key characteristics (a total of 1740), we found discrepancies in 200 (11.5%) instances. Furthermore, the comparison of the results between the register book and monthly reports was done in 200 reports for all fields except the result field, where the available data were only available for 94 fields. The comparison showed that there were 8 (8.5%) discrepancies in the test result field, 16 (8%) discrepancies in the X-ray field, 11 (5.5%) discrepancies in the sputum smear field, and 6 (3%) discrepancies in the fields of treatment plan and site of the disease.
| Field | Discrepancies (%) |
|---|---|
| Result of treatment** | 8 (8.5) |
| X-ray | 16 (8) |
| Sputum smear | 11 (5.5) |
| Treatment plan | 6 (3) |
| TB code | 5 (2.5) |
This is a result of review the available 145 treatment cards.
The results in the report that was sent to Ministry of Health were available for 94 patients only.
Discrepancies between register book and monthly reports, 2011, Saudi Arabia, Al-Madinah province.*
4. Discussion
The purpose of the PHS program is to provide information that helps public health workers and decision makers manage the disease and take appropriate actions. The ideal PHS system should have the following characteristics to derive the maximum benefit: the data should be accurate and complete and sent within the appropriate timeframe; the program should include all cases that occur and use a case definition and consistent lab protocols; the data should be analyzed and the feedback sent to those concerned; the decisions and implementation should be based on data; and the program should be easy to adapt [9].
The reporting of TB cases is required in KSA and the presence of high quality data is one of the critical requirements of the program. More accurate and complete data will increase the usefulness and utilization of information. High detection rates and accurate and complete data help health officials understand the trends of the disease in the country and its characteristics, as well as the burden. Moreover, the identification of new cases among patient contacts is one of the key steps in preventing TB’s spread. A strong PHS system helps decision makers establish and evaluate different interventions to control the disease. Furthermore, it assures appropriate medical treatment and follow-ups with patients. Moreover, the data can be used to make comparisons between different regions and other countries [10].
4.1. Underreporting
In our evaluation, we found underreporting of TB cases. In sum, 200 cases were reported and 244 cases were not reported; the sensitivity of the surveillance program was 45%. Most of the unreported cases were confirmed by laboratory testing (87.3%). Laboratories are fundamental components of TB PHS programs [8]. Laboratory results confirm the diagnosis and guide the treatment and contact investigation. Although data sent by laboratories do not contain clinical information, one study found that the reporting improved after the initiation of mandatory laboratory reporting [11].
Because the MOH cannot follow unreported cases, these patients have a higher risk of mortality and developing MDR-TB or even XDR-TB. Also, the contacts of those affected will not be screened for the disease, putting them at high risk of morbidity and mortality, especially if they have risk factors. Furthermore, under-notification will lead to underestimation of the disease burden and will limit the ability to plan for the appropriate prevention and control strategies.
Different factors could have led to the underreporting that was found. Physicians’ awareness about the notification requirements, procedures, and their importance and implications might have played a role in the underreporting. Lack of physician training in the notification process, pertaining to which diseases are notifiable, who receives the notifications, and how the notification is done, might have been another cause. In addition, physicians may have had time constraints that prevented them from reporting. The availability of the proper forms could have been another issue. In some cases, lack of laboratory confirmation may have led to underreporting [12].
4.2. Completeness of data
In the TB PHS program, most of the demographic, disease, and treatment data were complete. However, the test results data were not adequately completed. Accurate data are essential in the PHS system. Complete data will give precise information about disease trends, characteristics, and affected persons. With correct data, the potential risk factors can be examined and modified. Also, accurate data help program managers and decision makers implement interventions and evaluate them. The high incidence of incomplete test results data might have been due to lack of knowledge about how to fill out the reports. It may also have been due to physicians not being aware of the importance of each data field. They may have thought that this was just information collected and saved without being analyzed or used. Also, it may have been due to a lack of lab results or an inability to match the lab results with the right patients.
4.3. Contact investigation
Only smear-positive pulmonary TB patients can transmit the disease to contacts. As a result, the PHS program requires only the contacts of smear-positive pulmonary TB patients to be identified and investigated [13]. In the United States, the average number of contacts per patient is 10 [13]. In KSA, these data are not routinely gathered. In this study, we found the average number of identified contacts was 5 per patient. We also found 42 smear-positive, pulmonary TB patients whose contacts were not identified, and 20 patients whose 147 contacts were identified but not investigated. From this finding, we estimated the number of non-investigated contacts to be 210 (42 patients with an estimated five contacts each), plus 147 known non-investigated contacts, for a total of 357.
One of the essential steps in helping to control and eliminate TB is breaking down the disease’s infection chain by contact tracing. Undiagnosed patients infect their contacts; new patients will infect their contacts. As a result, the disease remains endemic. Different studies have shown contacts’ tracing and investigation to be an effective TB control strategy [14–17].
Many factors could have led to the neglect of contact investigations. There may have been weak communication between the regional coordinator and the health care center nearest to the patient’s home, which is responsible for doing the investigation. Also, the person assigned to investigate a contact may have been extremely busy or not have known the importance of the investigation and its benefits.
4.4. Identification
One of the challenges that we faced when collecting the data was the lack of consistency in the recording of people’s names. When the staff wrote names in Arabic, especially non-Saudi names, different spellings were used. Currently, the surveillance program assigns each patient a unique number that identifies the case. However, these numbers were not used on lab results and reporting forms. This led to duplication and the inability to match the lab results with the cases.
5. Recommendations and conclusion
5.1. Implementation of automated notification and reporting
The KSA TB program depends on paper-based notifications and monthly reports. Paper-based reporting is slow and incomplete [18]. Rapid and complete reporting is required to control and prevent TB. Different studies show that electronic reporting is faster and more complete [18–21]. Electronic reporting should include healthcare providers and laboratories. Because laboratories do not report detailed patient data, the electronic medical reporting system is imperative. Such a system would reduce the amount of incomplete data and delays in reporting that result from paper-based reporting. Also, it would improve the availability of detailed information from laboratory notification [18]. This system would also reduce the administrative time necessary for filling out paperwork.
5.2. Mandatory lab and suspected cases reporting
Because laboratories identified most of the missed cases, it is essential to implement mandatory laboratory reports. Also, all suspected cases should be reported to the regional coordinator even before lab confirmation. If a brief summary of suspected cases was provided, the regional coordinator could follow up on the cases through the hospitals and labs looking for either confirmation or an incorrect diagnosis. In addition, he/she could send the public health team to investigate the contacts early and prevent possible transmissions. As a result, the rate of underreported cases would be reduced and the sensitivity increased [22].
5.3. Investigation of the contacts
Identification and investigation of contacts is one of the most productive methods of detecting new cases at an early stage and preventing new transmissions. To improve this process, different strategies for the method of detection and evaluation should be used. Improving the communication between different health providers, especially in primary care centers, will facilitate progress [22].
5.4. Accurate and complete data
Accurate data are one of the key factors in a successful surveillance. The lack of consistency in the recording of names is a major problem in TB surveillance; there is neither a systematic way of recording names (i.e., first and last name, first and middle name only) nor standard spellings of non-Arabic names. Furthermore, the absence of an ID number in the lab results may lead to duplication or inability to match the lab results with the cases. The requirement of writing the ID number and the three names of the patients exactly as they are written on the patient’s ID card would prevent errors and duplication, help match all the patient papers, and, at the very least, lessen the time wasted by the regional coordinator trying to match them.
5.5. Other strategies
Other strategies include targeting physicians with a specific outreach program and ongoing training intended to raise their awareness and increase their response level. Also, revising the manual of the national TB control program, which contains disease information, reporting guidelines and control recommendations, would increase awareness and improve the notification rates. Periodic feedback from headquarters to coordinators and the dissemination of progress reports would raise the confidence of the providers in the program. In addition, periodic evaluation of health providers’ knowledge and attitudes regarding the program would help detect any weaknesses in the process. Finally, the program should be evaluated periodically to improve its quality and achieve optimum disease control.
Further study is needed to evaluate the time between diagnosis and notification, healthcare workers’ knowledge and practice in reporting TB cases, and the reasons for their not investigating all the patients’ contacts.
There were limitations of this study that prevented the evaluation of more characteristics of the program. First, all data were handwritten, which took a long time to read and evaluate. Second, the lab registrations were unclear and did not contain the whole names. Third, the treatment cards of some patients were not on hand at the coordinator’s office. Fourth, there were no other record systems of TB patients, so we could not apply the capture–recapture method. Finally, there were no electronic records of the inpatients in the hospitals, so we could not find the missed admitted cases.
In conclusion, our study revealed several areas in which TB surveillance data were lacking or inconsistent. Most critically, we found that over half of patients’ contacts were not identified or investigated. We also discovered a significant number of unreported cases, most of which were laboratory confirmed. In addition, there were varied rates of completeness for the different fields of TB notification reports, with the lab results and HIV test fields having the lowest rates of completion. In addition, we found a number of discrepancies between the treatment cards, logbooks, and the reported data.
Conflict of interest
None declared.
References
Cite this article
TY - JOUR AU - Mohammed J. Alkhalawi AU - Scott J.N. McNabb AU - Abdullah M. Assiri AU - Ziad A. Memish PY - 2015 DA - 2015/05/18 TI - Evaluation of tuberculosis public health surveillance, Al-Madinah province, Kingdom of Saudi Arabia, 2012 JO - Journal of Epidemiology and Global Health SP - 37 EP - 44 VL - 6 IS - 1 SN - 2210-6014 UR - https://doi.org/10.1016/j.jegh.2015.04.005 DO - 10.1016/j.jegh.2015.04.005 ID - Alkhalawi2015 ER -
