Safety and Efficacy of Haploidentical Peripheral Blood Stem Cell Transplantation for Myeloid Malignancies Using Post-transplantation Cyclophosphamide and Anti-thymocyte Globulin as Graft-versus-Host Disease Prophylaxis
Contributed equally to this publication
- DOI
- 10.2991/chi.d.190316.003How to use a DOI?
- Keywords
- Haploidentical hematopoietic cell transplantation; T-cell replete grafts; Post-transplant cyclophosphamide; Anti-thymocyte globulin; Myeloid malignancies; Graft-versus-host disease
- Abstract
Haploidentical stem cell transplantation (haploSCT) has greatly improved access to curative treatment for myeloid malignancies in patients without suitable matched sibling/unrelated donors. We investigated the safety and efficacy of haploSCT after reduced intensity conditioning (RIC) with anti-thymocyte globulin (ATG), post-transplant cyclophosphamide (PTCy), and cyclosporine to prevent rejection and graft-versus-host disease (GVHD). In this study, 47 patients received RIC using fludarabine, busulfan, and total body irradiation (200 cGy). Unmanipulated peripheral blood grafts were used. GVHD prophylaxis included ATG (4.5 mg/kg day−3 to −1), PTCy (50 mg/kg/day day +3, +4), and cyclosporine from day +5. The median follow-up was 15 months (range 3–27). Thirty one (66%) patients had acute myeloid leukemia (AML), 10 (21%) had high-risk myelodysplastic syndrome, and 6 (13%) had a myeloproliferative neoplasia. Median age was 60 years (range 22–73). The d+100 cumulative incidences of grade II–IV and III–IV acute GVHD were 17% (95% confidence interval (CI) 7.9–29.1) and 6.4% (1.6–15.9), respectively. The cumulative incidence of moderate-severe chronic GVHD at 1 year was 15.2% (95% CI 6.5–27.1). Overall survival (OS) and relapse-free survival (RFS) were 55.2% (95% CI 39.5–68.4) and 49.5% (95% CI 34.2–63), respectively. Nonrelapse mortality (NRM) for all patients at 1 year was 37.1% (95% CI 23.2–51.1). Infection was the main cause of death (26%). For AML, 1-year OS, RFS, and NRM were 64.1% (95% CI 43.3–78.9), 54.5 (95% CI 34.6–70.7), and 26.8% (95% CI 12.3–43.6), respectively. In conclusion, unmanipulated haploidentical peripheral blood stem cells (PBSC) transplantation following RIC and dual in vivo T-cell depletion results in a low incidence of acute and chronic GVHD for patients diagnosed with myeloid malignancies.
- Copyright
- © 2019 International Academy for Clinical Hematology. Publishing services by Atlantis Press International B.V.
- Open Access
- This is an open access article distributed under the CC BY-NC 4.0 license (http://creativecommons.org/licenses/by-nc/4.0/).
1. INTRODUCTION
Allogeneic stem cell transplantation (alloHSCT) is potentially curative for some high-risk hematologic cancers [1]. When available, human leukocyte antigen (HLA)-matched related donors (MRD) are the preferred donor sources. However, only approximately 30% of patients from Western countries have compatible sibling donors who are fit to donate stem cells [2]. The increase in matched unrelated donor (MUD) registries has improved donor availability for many patients, although this is less certain for ethnic minorities [2,3].
Haploidentical donors are increasingly chosen as alternatives when HLA-matched related and unrelated donors are unavailable [2,3]. This reduces the time to transplantation, expenses, and gives access to a reliable source of donor cells to salvage failing grafts or treat relapse. A virtually immediate access to a haploidentical donor is of significant benefit, particularly in the case of patients with acute myeloid leukemia (AML), where delay in transplantation due to donor availability can be catastrophic [4].
The optimal choice of conditioning regimens and graft-versus-host disease (GVHD) prophylaxis in haploidentical stem cell transplant (haploHSCT) varies greatly according to center preference. Several regimens for GVHD prophylaxis in haploHSCT are based on in vivo T-cell depletion with post-transplant cyclophosphamide (PTCy) [5–7]. The safety and efficacy of the use of post-transplant high dose PTCy combined with T-cell replete grafts in haploHSCT have been reported by several recent studies [7–10]. The use of anti-thymocyte globulin (ATG) as part of conditioning regimens leads to more rapid achievement of complete donor chimerism, in particular when using alternative donors [11,12].
Since August 2016, we have used a reduced intensity conditioning (RIC) regimen of fludarabine (Flu), busulfan (Bu), and low-dose total body irradiation (TBI) as standard of care for all eligible haploHSCT patients at the Princess Margaret Hospital, Toronto. Rabbit-ATG with PTCy and cyclosporine (CsA) are used for GVHD prophylaxis [13]. Herein, we analyze the safety and efficacy of this approach in haploHSCT for myeloid malignancies.
2. PATIENTS AND METHODS
2.1. Patient Selection
Between August 2016 and August 2018, 47 adults with myeloid hematologic malignancies underwent haploHSCT at the Princess Margaret Cancer Center, Toronto, Canada. All received identical RIC with rabbit ATG, PTCy, and CsA to prevent GVHD and graft rejection. All were transplanted with unmanipulated T-cell replete peripheral blood stem cells (PBSC) grafts. The median follow-up of patients was 15 months (range 3–27). The present study was conducted in accordance with the Declaration of Helsinki. As per institutional guidelines, it was reviewed and approved by the Ethics Committee at the Princess Margaret Cancer Centre, Toronto, Canada. All patients/donors provided informed consent.
2.2. Conditioning, Immunosuppression, and Supportive Care
The institutional RIC regimen consisted of Flu (30 mg/m2 × 4 days from days −5 to −2) with Bu (3.2 mg/kg × 2 days from days −3 to −2) and 200 cGy of TBI for all eligible patients receiving haploHSCT [13]. To prevent graft rejection and for GVHD prophylaxis, ATG (Thymoglobulin; Genzyme-Sanofi, Lyon, France) (cumulative dose 4.5 mg/kg), combined with PTCy (50 mg/kg × 2 days from days +3 to +4) and CsA, starting at 2.5 mg/kg IV q12h on day +5 and modified to achieve therapeutic levels between 200 and 400 mcg/L, were used. CsA was tapered around day +45 to +60 for patients who did not develop acute GVHD. Daily G-CSF 300 mcg subcutaneously was given from day +7 until neutrophils reached >1.5 × 109/L. Ursodeoxycholic acid (UDCA) at 500 mg p.o. twice daily was used for prevention of hepatic complications. Infection prophylaxis included ciprofloxacin until neutrophil engraftment; caspofungin/micafungin until neutrophil engraftment followed by posaconazole for 3 months; acyclovir; and either cotrimoxazole or inhaled pentamidine.
2.3. Engraftment, Chimerism Analysis, and Disease Monitoring
Engraftment was defined as an absolute neutrophil count ≥0.5 × 109/L on the first of 3 consecutive days. Platelet recovery was defined as transfusion-independent platelet counts maintained >20 × 109/L on the first of three consecutive days. Donor total chimerism was assessed in whole blood by polymerase chain reaction for short tandem repeat polymorphisms at day +30 and +60. Bone marrow examinations were performed for all patients at day +60 to assess marrow cellularity and to monitor disease status.
2.4. Statistical Methods
Clinical variables, outcome, and complications were collected through retrospective chart review. Data were updated in December 2018. Baseline characteristics were reported using descriptive statistics. The main variables of interest were overall survival (OS), relapse-free survival (RFS), nonrelapse mortality (NRM), and the cumulative incidences of GVHD and relapse. OS and PFS were calculated using the Kaplan–Meier product-limit method and the impact of variables was assessed using the Log-rank test. NRM and cumulative incidence of relapse (CIR) were estimated using relapse as a competing risk (Fine and Gray analysis) and assessed at 100 days post-alloHSCT. Acute and chronic GHVD rates were estimated using the cumulative incidence method considering death and relapse as competing risks. Cox proportional hazards regression was used to assess the impact of covariates of interest to survival outcome. All P values were two-sided and, for the statistical analyses, P<0.05 was considered to indicate a significant result. Statistical analysis was performed using STATA and EZR software (Saitama Medical Center, Jichi Medical University, Saitama, Japan). EZR is a modified version of R commander (version 2.12.1) (http://www.jichi.ac.jp/saitama-sct/SaitamaHP.files/statmedEN.html) [14].
3. RESULTS
3.1. Patient Information
Baseline patient and donor characteristics are summarized in Table 1. The outcomes of 47 patients were analyzed. This included 31(66%) with AML, 10 (21%) with high-risk myelodysplastic syndrome (MDS), and 6 (13%) with myeloproliferative neoplasia (MPN). Among the AML patients, 4 (13%) had favorable cytogenetics, 19 (61%) normal karyotype, and 8 (26%) had complex/high risk cytogenetic changes at diagnosis. Twenty two (74%) had primary AML, five (16%) secondary AML (two after MPN; four on a background of MDS), and two (10%) were diagnosed with therapy-related AML. At the time of the transplant, 27 (88%) recipients were in a first complete remission (CR), 4 of them after 2 lines of induction chemotherapy. Two (6%) each were in a second and third CR. Of the 10 (21%) patients with MDS, half had complex cytogenetics at diagnosis. Two (20%) had therapy-related MDS. Prior to transplant, seven were treated with azacitidine and two received induction chemotherapy to reduce the blast count to <10%. One (10%) patient proceeded to transplant without pretreatment. Among MPN patients, five (83%) were diagnosed with primary myelofibrosis and one (17%) case had post-polycythemia vera myelofibrosis. All patients received prior therapy with ruxolitinib.
| Overall (N = 47) n (%) | AML (N = 31) n (%) | MDS (N = 10) n (%) | MPN (N = 6) n (%) | |
|---|---|---|---|---|
| Patient Clinical Parameters | ||||
| Recipient age (years) median (range) | 60 (22–73) | 56 (22–73) | 67 (44–72) | 63 (47–67) |
| ≥60 years | 25 (53) | 12 (39) | 9 (90) | 4 (68) |
| Sex | ||||
| Female | 21 (45) | 15 (48) | 3 (30) | 3 (50) |
| Disease risk index | ||||
| Low | 0 | 0 | 0 | 0 |
| Intermediate | 31 (66) | 21 (68) | 5 (50) | 5 (83) |
| High | 13 (28) | 9 (29) | 3 (30) | 1 (17) |
| Very high | 3 (6) | 1 (3) | 2 (20) | 0 |
| Disease status | ||||
| Primary disease | 36 (77) | 23 (74) | 8 (80) | 5 (83) |
| Secondary disease | 6 (13) | 5 (16) | 0 | 1 (17) |
| Therapy-related | 5 (10) | 3 (10) | 2 (20) | N/A |
| Stage at transplant | ||||
| CR1 | 34 (73) | 27 (88) | 7 (70) | - |
| CR2 | 2 (4) | 2 (6) | 0 | - |
| CR3 | 2 (4) | 2 (6) | 0 | - |
| Active disease / partial response | 9 (19) | 0 | 3 (30) | 6 (100) |
| HCT-CI | ||||
| ≥3 | 20 (43) | 10 (32) | 6 (60) | 4 (66) |
| KPS | ||||
| ≤80 | 6 (13) | 5 (16) | 1 (10) | 0 |
| HLA mismatch | ||||
| 8/10 | 3 (6) | 2 (6) | 0 | 1 (17) |
| 7/10 | 2 (4) | 2 (6) | 0 | 0 |
| 6/10 | 6 (13) | 2 (6) | 1 (10) | 3 (50) |
| 5/10 | 36 (77) | 25 (82) | 9 (90) | 2 (33) |
| Donor and Graft Parameters | ||||
| Donor age (years) median (range) | 38 (11–62) | 37 (11–62) | 39 (26–46) | 35 (27–51) |
| ≥50 years | 9 (19) | 13 (42) | 0 | 1 (17) |
| Donor gender | ||||
| Female | 17 (36) | 14 (45) | 2 (20) | 1 (17) |
| Fresh/cryopreserved graft | ||||
| Cryopreserved | 28 (60) | 20 (65) | 5 (50) | 3 (50) |
| CMV immune status (Donor–Recipient) | ||||
| Negative / positive | 7 (15) | 5 (16) | 2 (20) | 0 |
| Other | 40 (85) | 26 (84) | 8 (80) | 6 (100) |
Abbreviations: AML: Acute myeloid leukemia, MDS: Myelodysplastic syndrome, MPN: Myeloproliferative neoplasm, HCT-CI: Hematopoietic cell transplant-comorbidity index, KPS: Karnofsky performance status, HLA: Human leukocyte antigen, CMV: Cytomegalovirus, N/A: None applicable.
Clinical characteristics of patients and donors.
3.2. Pretransplant, Donor, and Graft-Related Information
The median age of all 47 patients was 60 years (range 22–73), and 25 (53%) of them were ≥60 years. Six (13%) had a Karnofsky performance status (KPS) ≤80 and 20 (43%) had a hematopoietic cell transplant comorbidity index score ≥3 (Table 1). The median donor age was 37 years (range 11–62). Thirty donors (64%) were male. Two-thirds were mismatched at five HLA loci, and 17 (36%) were cytomegalovirus (CMV) mismatched. Cryopreserved PBSC grafts were given to 28 (60%) recipients, whereas 16 (40%) received fresh products. A median of 9.48 × 106 CD34+cells/kg (range 3.73–28.60) recipient body weight stem cells were collected and consecutively infused. Donor-specific anti-HLA antibodies (DSAs) were assessed prior to transplant and were negative in all recipients.
3.3. Post-transplant Information and Complications
Forty-four (94%) patients successfully engrafted after haploHSCT. The median time to neutrophil engraftment was 16 days (range 8–43), and to platelet engraftment was 19 days (7–217). Thirty-nine (83%) patients reached chimerism levels >95% donor at day +30. Graft failure (GF) was documented in 10 (21%) patients; of these, 4 had primary GF and 6 secondary GF. A second transplant was done in four patients and two of them successfully engrafted and clinically recovered. Post-transplant complications are summarized in Table 2. Briefly, the median duration of the transplant admission was 32 days (range 6–87). Thirty-eight (81%) patients developed mucositis, which was grade 3–4 in only 5 (10%). Six (13%) patients had SOS, with one case of grade 3; all instances were managed with diuretics and responded to treatment. Engraftment syndrome was documented in two (4%) patients and was successfully treated with steroids. Sixteen (34%) patients had transient cardiac complications during hospitalization: nine recipients had fluid overload that responded to diuretics, one developed atrial fibrillation, and three had pericardial complications (one pericardial effusion, one myopericarditis, and one pericarditis). All symptoms resolved without sequelae. Finally, 10 (21%) recipients had nephrotoxicity but only one had grade 3 acute kidney injury, which resolved with conservative management.
| Overall (N = 47) n (%) | AML (N = 31) n (%) | MDS (N = 10) n (%) | MPN (N = 6) n (%) | |
|---|---|---|---|---|
| Sinusoidal obstruction syndrome | ||||
| All Grades | 6 (13) | 2 (6) | 1 (10) | 3 (50) |
| Grade 3–4 | 1 (2) | 1 | 0 | 0 |
| Engraftment syndrome | 2 (4) | 2 (6) | 0 | 0 |
| >95% chimerism at day +30 | 39 (83) | 30 (97) | 6 (60) | 3 (50) |
| Graft failure | 10 (21) | 3 (10) | 5 (50) | 2 (33) |
| Primary | 4 (9) | 0 | 2 (20) | 2 (33) |
| Secondary | 6 (13) | 3 (10) | 3 (30) | 0 |
| Infections | ||||
| Febrile neutropenia | 38 (81) | 26 (83) | 9 (90) | 3 (50) |
| Blood cultures positive (bacteremia) | 17 (36) | 13 (42) | 4 (40) | 0 |
| CMV reactivation | 36 (77) | 22 (71) | 10 (100) | 4 (67) |
| CMV disease (probable or confirmed) | 4 (9) | 3 (10) | 1 (10) | 0 |
| Epstein–Barr virus reactivation | 29 (62) | 21 (68) | 5 (50) | 3 (50) |
| Presumed PTLD | 2 (4) | 2 (6) | 0 | 0 |
| Biopsy-proven PTLD | 3 (6) | 3 (10) | 0 | 0 |
| Patients treated with rituximab | 5 (11) | 5 (16) | 0 | 0 |
| Clinical response | 100% | 100% | 0 | 0 |
| BK viruria | 12 (26) | 8 (26) | 2 (20) | 2 (33) |
| Other viral infections | 14 (30) | 11 (35) | 3 (37) | None |
| Coxsackie infection | 1 | 1 | 0 | |
| Herpes virus type 1 | 6 | 4 | 2 | |
| Influenza A | 3 | 3 | 0 | |
| Parainfluenza | 1 | 1 | 0 | |
| Coronavirus | 1 | 1 | 0 | |
| Varicella zoster | 1 | 1 | 0 | |
| Respiratory syncytial virus | 1 | 0 | 1 | |
| Fungal infection | ||||
| Probable | 6 (13) | 4 (13) | 2 (25) | 0 |
| Proven | 0 | 0 | 0 | |
| Outcomes | ||||
| Relapse | 8 (17) | 5 (16) | 2 (20) | 1 (16) |
| Death | 22 (47) | 12 (39) | 8 (80) | 2 (33) |
| Causes of death | ||||
| Relapse | 2 (4) | 2 (6) | 1(10) | 0 |
| Infection | 11 (23) | 8 (26) | 3 (30) | 0 |
| Graft failure | 7 (15) | 1 (3) | 4 (40) | 2 (33) |
| Graft-versus-host disease | 0 | 0 | 0 | 0 |
| Multiorgan failure | 2 (4) | 2 (6) | 0 | 0 |
Abbreviations: AML: Acute myeloid leukemia, MDS: Myelodysplastic syndrome, MPN: Myeloproliferative neoplasm, PTLD: Post-transplant lymphoproliferative disorder, CMV: Cytomegalovirus.
Post-transplant complications.
3.4. Infectious Complications
Infectious complications are summarized in the Table 2. CMV reactivated in 36 (77%) patients. Reactivations were managed with ganciclovir or valganciclovir and reduction of immunosuppression, as appropriate. Four (9%) recipients developed probable or confirmed CMV disease, two of them pulmonary and two gastrointestinal involvement. All four were treated with intravenous ganciclovir therapy and a clinical response was documented in two (50%) cases.
Quantitative blood tests for Epstein–Barr virus (EBV) replication were positive in 29 (62%) recipients. Three (6%) developed a post-transplant lymphoproliferative disorder (PTLD) which was confirmed on biopsies of the affected areas. Two (4%) recipients required preemptive rituximab due to fever and EBV viremia >1 × 106 copies/mL. PTLD was treated by a rapid taper of systemic immunosuppression, accompanied by rituximab (375 mg/m2 once weekly) for a maximum of four doses. Complete responses with clearance of viremia and regression of affected nodal or extranodal sites were documented in all patients. BK viruria was documented in 12 (26%) recipients and other viral infections in 14 (30%) recipients. Finally, probable invasive fungal infection (evidenced by positive galactomannan in broncho-alveolar lavage fluid with consistent radiological findings) was seen in six (13%) post-transplant. One patient succumbed to complications of fungal pneumonia.
3.5. Graft-versus-Host Disease
Acute and chronic GVHD were graded according to the Keystone criteria and the National Institutes of Health Chronic Graft-versus-Host Disease Consensus, respectively [15,16]. The cumulative incidence of grade 2–4, and grade 3–4 acute GVHD at d + 100 was 17% (95% interval confidence (IC) 7.9–29.1) and 6.4% (1.6–15.9), respectively (Table 2). Overall, acute GVHD was identified in 22 (47%) patients. Twelve (26%) recipients developed grade 1 acute GVHD, six (13%) grade 2, and four (9%) developed grade 3 acute GVHD. No patient developed grade 4 acute GVHD. Skin (38%) was the most frequently involved organ followed by the gut (19%) and liver (13%). All patients were treated with immunosuppressive therapy consisting of steroids and calcineurin inhibitors with good clinical responses. No cases of steroid-refractory acute GVHD were seen. The cumulative incidence of moderate and severe chronic GVHD at 6 months and 1 year was 13% (95% IC 5.2–24.4) and 15.2% (95% 6.5–27.1), respectively. Chronic GVHD of any grade was seen in eight (17%) recipients. Mild GVHD was diagnosed in one (2%) recipient and moderate GVHD was seen in seven (15%). No cases of severe GVHD were observed. Seven (15%) patients with moderate chronic GVHD required systemic immunosuppression and demonstrated appropriate responses. Supplementary Figs. S1 to S4 show the cumulative incidence of acute and chronic GVHD, and relapse for all 47 patients.
3.6. Relapse and Outcomes
Eight (17%) patients relapsed after haploHSCT. This included six with AML, one with high-risk MDS, and one with primary myelofibrosis. With a median follow up of 15 months (range 3–37), 22 (47%) deaths were documented. The causes of death were as follows: 11 (23%) secondary to an infection, 7 (15%) secondary to GF, 2 (4%) relapse, and 2 (4%) due to multiorgan failure. Survival information is summarized in Tables 3 and 4. One-year OS was 54.1% (95% confidence interval [CI] 38.1–67.6). One-year RFS was 48.2% (95% CI 32.8–62). For the entire cohort, the NRM at 1 year was 37.1% (95% CI 23.2–51.1) and the CIR at 1 year was 13.2% (95% CI 5.3–24.9). AML patients comprised the largest subgroup (66%) and had 1-year OS and RFS of 64.1% (95% CI 43.3–78.9) and 54.5 (95% CI 34.6–70.7), respectively (Fig. 1). For AML patients, the 1-year NRM and CIR were 26.8% (95% CI 12.3–43.6) and 20.4% (95% CI 8–36.7), respectively (Table 3).
| Overall N = 47 % (95% CI) | AML (N = 31) % (95% CI) | |
|---|---|---|
| Overall survival | ||
| 6 months | 68.9 (53.2–80.3) | 75.9 (55.9–87.7) |
| 1 year | 54.1 (38.1–67.6) | 64.1 (43.3–78.9) |
| Relapse-free survival | ||
| 6 months | 60.6 (45–73.1) | 66.3 (46.3–80.3) |
| 1 year | 48.2 (32.8–62) | 54.5 (34.6–70.7) |
| Nonrelapse mortality | ||
| 6 months | 30.2 (17.6–43.7) | 23 (9.9–39.3) |
| 1 year | 37.1 (23.2–51.1) | 26.8 (12.3–43.6) |
| Cumulative incidence of relapse | ||
| 6 months | 8.6 (2.7–18.8) | 13 (4–27.6) |
| 1 year | 13.2 (5.3–24.9) | 20.4 (8–36.7) |
Abbreviations: AML: Acute myeloid leukemia, CI: Confidence interval.
Outcomes: Overall survival, relapse-free survival, nonrelapse mortality, and cumulative incidence of relapse.
| 1 year OS % (95% CI) | P value* | 1 year RFS % (95% CI) | P value* | |
|---|---|---|---|---|
| Recipient age | ||||
| <65 | 65.6 (40.8–82) | 0.227 | 52.3 (29.3–71) | 0.698 |
| ≥65 | 45.2 (24.7–63.7) | 45.6 (25.1–64) | ||
| Disease risk index | ||||
| Intermediate and high | 58.8 (42.2–72.1) | 0.028 | 52.5 (36.3–66.3) | 0.082 |
| Very high | 0 | 0 | ||
| Karnofsky performance status | ||||
| <80 | 53.1 (36.1–67.5) | 0.991 | 46.3 (30.1–61) | 0.653 |
| ≥80 | 66.6 (19.4–90.4) | 66.6 (19.4–90.4) | ||
| HCT-CI | ||||
| <4 | 63.3 (44.3–77.4) | 0.224 | 58.1 (39.5–72.8) | 0.188 |
| ≥4 | 33.3 (10.2–58.8) | 25 (6–50.4) | ||
| Donor sex | ||||
| Male | 62.2 (42.1–77) | 0.116 | 53.1 (34–69) | 0.529 |
| Female | 42.3 (18–64.9) | 42.3 (18–64.9) |
Univariate analysis; Log-rank test.
Abbreviations: OS: Overall survival, RFS: Relapse-free survival, HCT-CI: Hematopoietic cell transplantation-comorbidity index, CI: Confidence interval.
Univariate analysis of risk factors for overall survival and relapse-free survival.
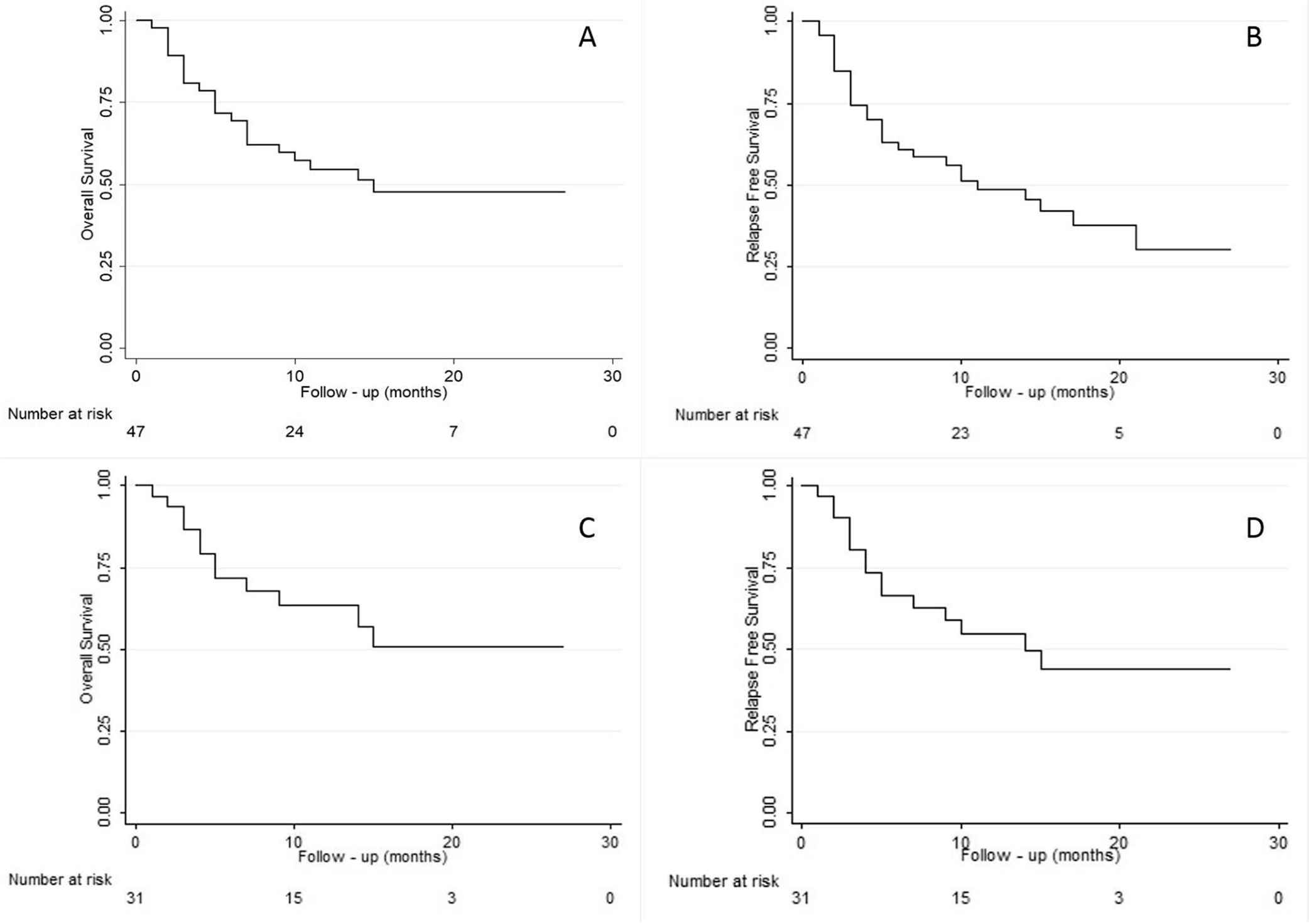
(A) Overall survival and (B) relapse-free survival for all patients (N = 47); (C) overall survival and (D) relapse-free survival for AML patients (n = 31).
Very high-risk patients assessed by the disease risk index tool designed by the Center for International Blood and Marrow Transplant Research (CIBMTR) had a significantly worse OS (P = 0.028). Recipient age >65 years, KPS ≤80% prior to transplant, or HCT-CI ≥4 did not have a significant impact on OS or RFS (Table 4 and Fig. 2).
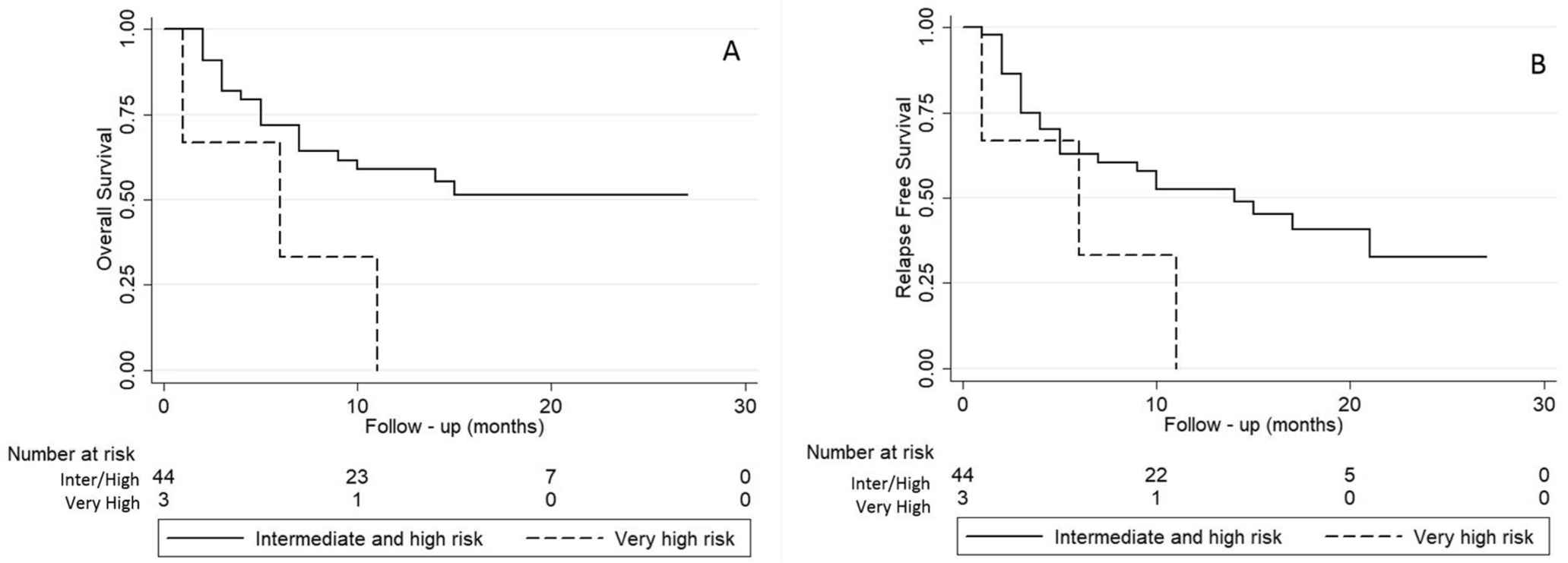
(A) Overall survival and (B) relapse-free survival for all patients (N = 47) stratified according to disease risk index.
4. DISCUSSION
HaploHSCT is a suitable alternative for patients diagnosed with high-risk hematologic malignancies in the absence of a matched sibling or unrelated donor [10]. When used with unmanipulated-T-cell replete PBSC grafts with ATG, PTCy, and calcineurin inhibitors results in a very low rate of acute and chronic GVHD in these patients [13]. While this combination was first described by our group in patients with a variety of hematologic neoplasms (including lymphoid), this report focuses on myeloid malignancies, predominantly AML.
The combination of PTCy and ATG seems to ameliorate the increased risk of acute and clinically significant chronic GVHD in the context of PBSC grafts. Cyclophosphamide in this setting selectively targets and induces apoptosis in alloreactive T-cells which ordinarily proliferate early after transplant and can cause GVHD. Furthermore, it spares regulatory T-cells (Tregs) and preserves quiescent hematopoietic stem and other progenitor cells [7–10]. The addition of ATG to standard conditioning has allowed better engraftment and rapid achievement of complete donor chimerism, particularly with alternative donors [11,12]. This benefit is somewhat offset by delayed immunological recovery and, thereby, an increased risk of infections, in particular those due to CMV and EBV [11,12].
There are several reports on the applicability of RIC for haploSCT in myeloid malignancies [12,13,17]. The RIC protocol described here is a low-toxicity regimen, which induces immunosuppression to a degree sufficient to permit consistent engraftment despite the HLA-haplotype barrier [12,13]. The overall intensity of the conditioning regimen may be more potent than the sum of its parts, due to the combination of Flu and Bu with PTCy, thereby reducing the relapse risk [12,18].
HaploSCT recipients have an increased risk of infectious complications due to the higher degree of immunosuppression [6]. Infection was the most frequent cause of death in our study. Despite the use of T-cell replete grafts, the degree of in vivo T-cell depletion appears to be considerable in its impact on infection rates. To ameliorate this effect while preserving the GVHD preventing properties of this regimen, we have since reduced the dose of ATG to 2 mg/kg.
The rate of CMV and EBV reactivation was high. All patients were monitored in order to promptly detect CMV and EBV DNA-emia. Although there was a high incidence of CMV reactivation, relatively few patients developed CMV disease with only one refractory case. The use of antiviral agents such as ganciclovir and valganciclovir are effective but can compromise graft function, leading to prolonged cytopenias or GF. Optimal preventive strategies for CMV reactivation in the context of haploSCT are lacking. Prophylactic approaches using novel therapeutic agents such as letermovir are being studied and may lead to safer alternatives [19,20].
EBV reactivations were seen in 62% cases and EBV-related PTLD was identified in 10%. No patient died of PTLD, and complete responses were seen in all treated with rituximab. EBV-infected B cells are controlled by EBV specific cytotoxic T-cell lymphocytes. Increased immunosuppression and the degree of T-cell depletion affect the risk of developing PTLD [21,22]. The high rate of EBV-reactivation/PTLD observed in this cohort is most likely due to the use of ATG. Furthermore, the rate of detection of EBV reactivation could be higher, due to consistent weekly EBV monitoring by quantitative PCR. EBV-matched donor selection may decrease the incidence of EBV reactivation, and the use of preemptive rituximab may reduce the development of PTLD. As with CMV, refinements in the dose of ATG may decrease the infection rate [21,22].
HaploHSCT is associated with a higher rate of GF secondary to alloreactive reactions related to major HLA mismatch between donor and recipient [23,24]. The reported incidence of GF in haploHSCT ranges from 10% to 20% [25,26]. Higher rate of engraftment has been described with the infusion of larger doses of hematopoietic stem cells [26]. In our cohort, the minimum requested dose was 4 × 106 CD34+ cell/kg, and the median infused cell dose was 9.48 × 106 CD34+ cells/kg. A high rate of GF was seen in our cohort. Graft rejection in haploSCT may be attributed to the effects of a cytolytic host-versus-graft reaction mediated by host T and/or natural killer (NK) cells that persist after conditioning. Primary GF was seen in four (9%) recipients. This high rate could be secondary to a host immunologic reaction against donor cells. The underlying disease may also play a role, as half of these patients had MPN. No patient developed DSAs after the primary GF. Secondary GF was diagnosed in six (13%) recipients. In all, this occurred following severe infectious complications, including viral reactivations requiring prolonged treatment with antiviral therapy. It is hypothesized that repeated external stress on a relatively fragile graft in the early post-transplant phase might compromise its survival and lead to secondary GF. Less toxic alternatives to existing antiviral therapies such as letermovir, or the use of novel approaches like virus specific T-cells may address this issue in future.
HaploHSCT shows great promise in the management of hematologic malignancies, particularly in patients without access to matched sibling and unrelated donors. Despite major advances, acute and chronic GVHD have hampered survival and increased morbidity and mortality. The combination of ATG and PTCy has been shown to be effective in reducing both acute and chronic GVHD to a large extent, albeit at the expense of higher infection rates. Further refinements are undoubtedly required to improve outcomes and reduce complications. Finally, the use of innovative post-transplant disease control strategies such as maintenance therapy or preemptive donor lymphocyte infusions may be appropriate for the AML patients at high risk of relapse.
SUPPLEMENTARY FIGURES
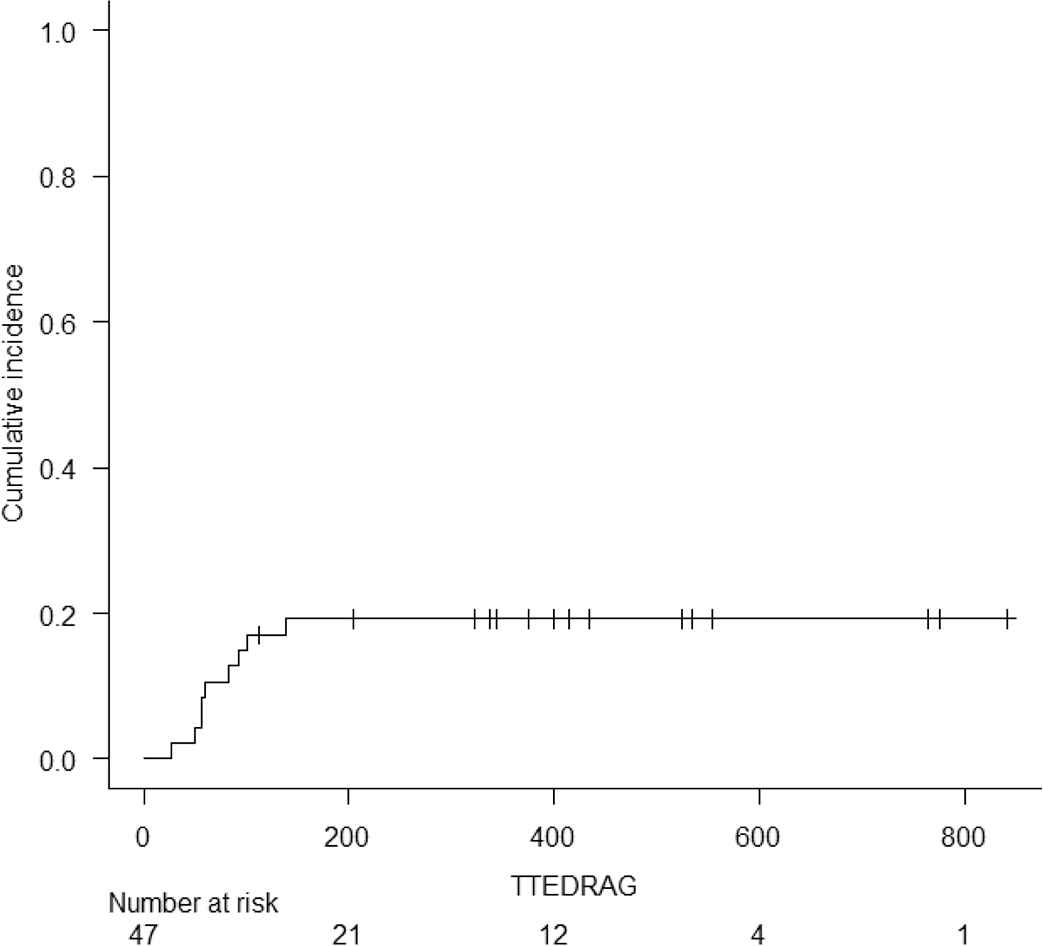
Cumulative incidence of Grade II to IV Acute Graft versus Host Disease
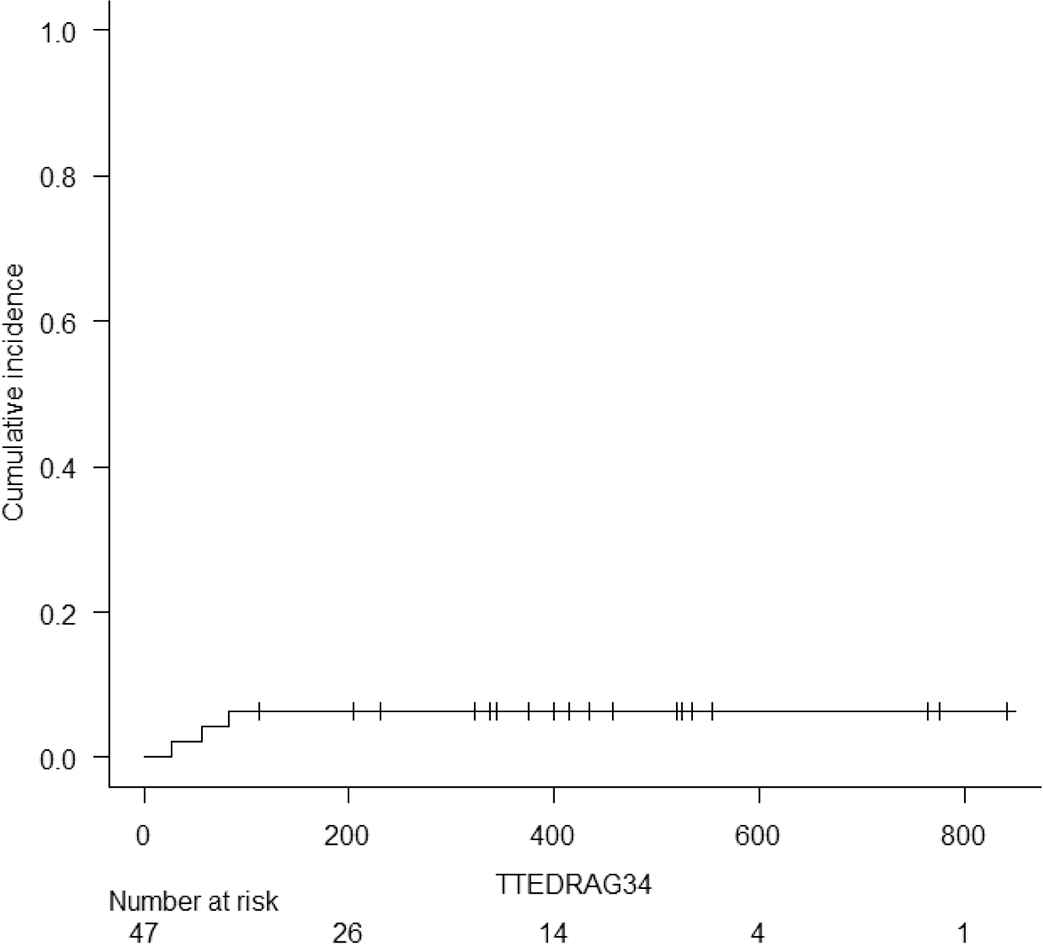
Cumulative incidence of Grade III to IV Acute Graft versus Host Disease
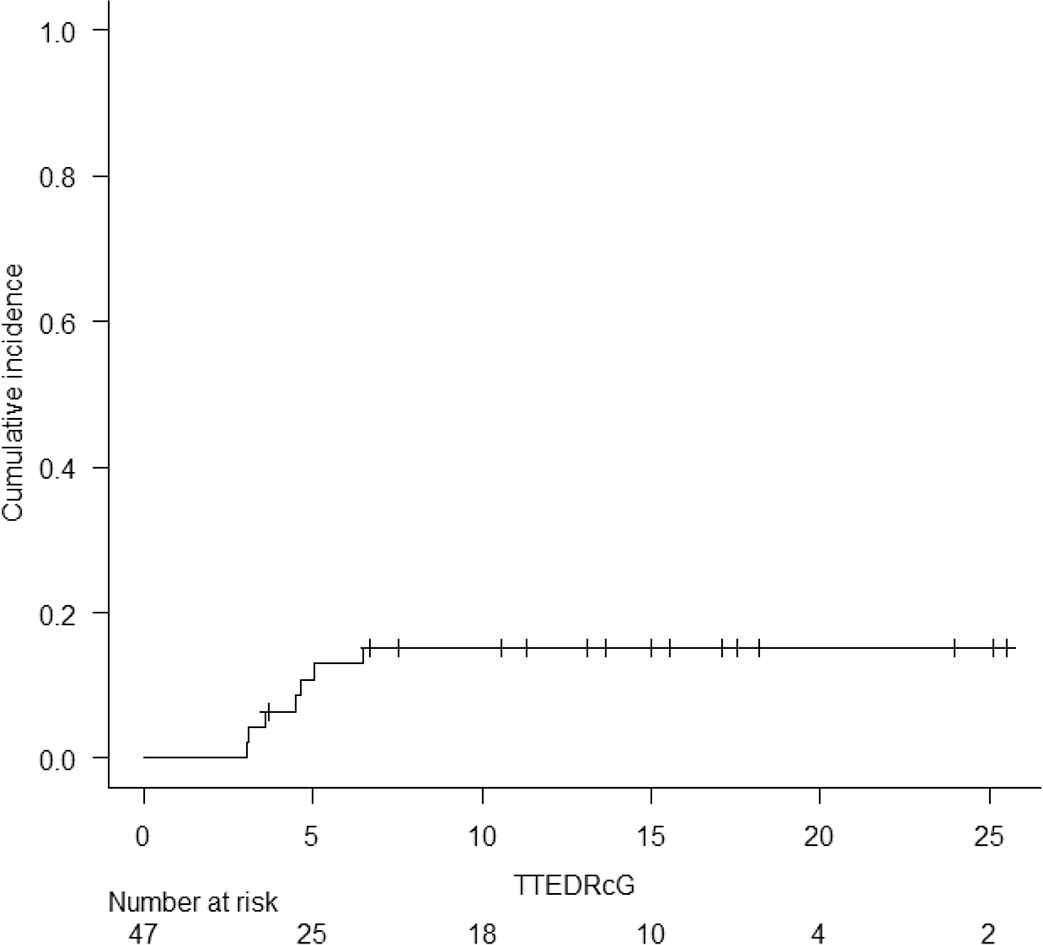
Cumulative incidence of Moderate/Severe Chronic Graft versus Host Disease
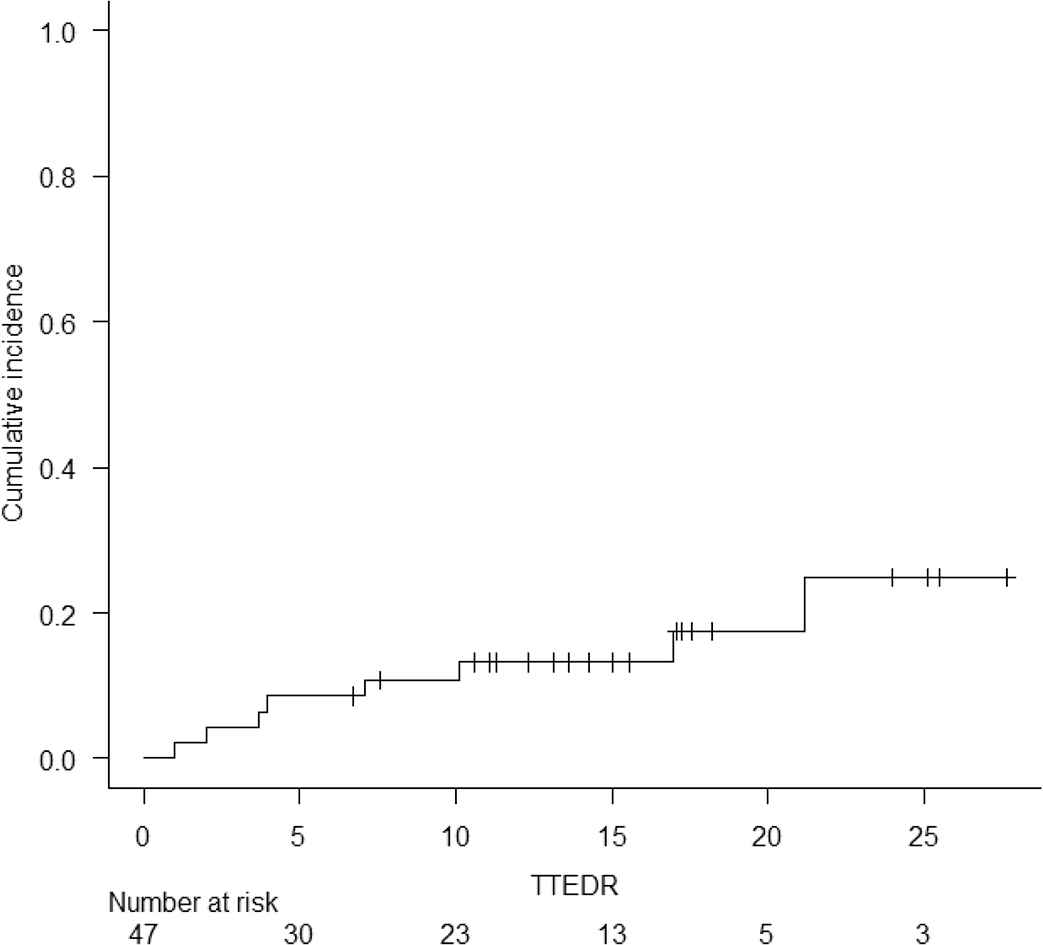
Cumulative incidence of Relapse
CONFLICT OF INTEREST
The authors declare no relevant conflicts of interest, financial or otherwise.
Funding
There are no external funding sources to declare.
ACKNOWLEDGMENT
QS and AL collected and analyzed the data and contributed equally to the writing of this paper. WL, ZA, DL, DK, FM, ST, RK, JL, JM, and AV managed patients, provided valuable input into the study design, analysis, and interpretation and reviewed the manuscript.
Footnotes
REFERENCES
Cite this article
TY - JOUR AU - Maria Queralt Salas AU - Arjun Datt Law AU - Wilson Lam AU - Zeyad Al-Shaibani AU - David Loach AU - Dennis (Dong Hwan) Kim AU - Fotios V. Michelis AU - Santhosh Thyagu AU - Rajat Kumar AU - Jeffrey Howard Lipton AU - Jonas Mattsson AU - Auro Viswabandya PY - 2019 DA - 2019/06/11 TI - Safety and Efficacy of Haploidentical Peripheral Blood Stem Cell Transplantation for Myeloid Malignancies Using Post-transplantation Cyclophosphamide and Anti-thymocyte Globulin as Graft-versus-Host Disease Prophylaxis JO - Clinical Hematology International SP - 105 EP - 113 VL - 1 IS - 2 SN - 2590-0048 UR - https://doi.org/10.2991/chi.d.190316.003 DO - 10.2991/chi.d.190316.003 ID - Salas2019 ER -
